
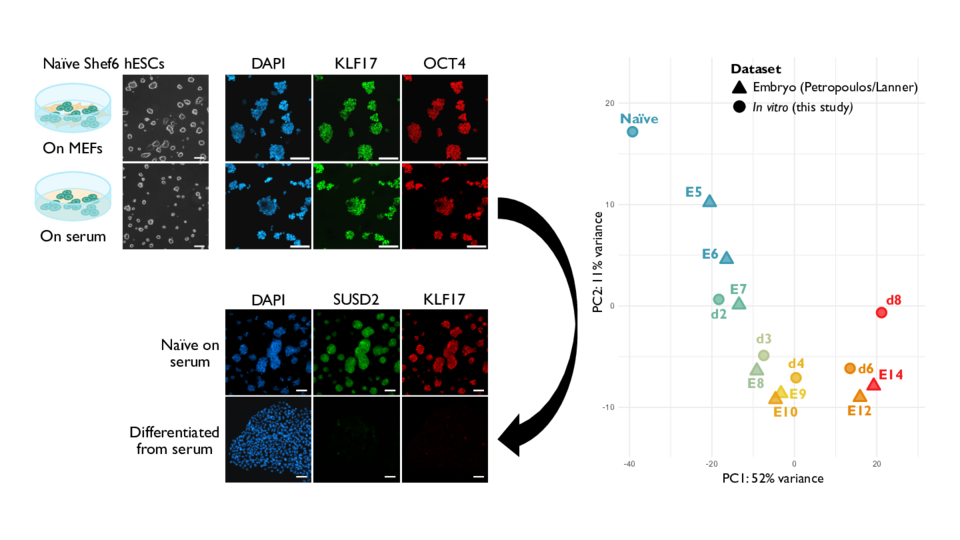
Human pluripotent stem cells (hPSCs) can self-renew indefinitely and differentiate into almost any cell type in the body. Capturing them in their most primitive, naïve state, however, has been notoriously difficult. “The only way to capture truly naïve human pluripotent cells required co-culturing with mouse feeder cells, which is conceptually, practically, and medically problematic,” explains group leader Martin Leeb. Notwithstanding the fact that producing mouse feeder cells is extremely time consuming and commercial alternatives are prohibitively expensive, they also complicate experiments, limit scalability, and pose clear challenges for translational applications.
Instead of engineering a completely new solution, the researchers turned to fetal calf serum, one of the oldest and most widely used components in cell culture. By systematically optimizing it as a surface coating, the scientists found that serum alone could support the long-term growth of naïve hPSCs. Molecular, transcriptomic, and differentiation analyses confirmed that the cells remained developmentally authentic. As first author Michael Oberhuemer puts it: “When we induce differentiation, the cells follow almost exactly the same molecular trajectory we see in the human embryo.”
A defining strength of the study lies in its collaborative design. “This is not a one-lab method paper – multiple independent labs have shown that the cells are molecularly indistinguishable from feeder-cultured cells”, says Martin. The study builds on an initial concept from the Martello lab (University of Padua), was co-led by the Zylicz lab (University of Copenhagen), and included contributions from the Rivron lab (IMBA). Together, these expert stem cell labs tested the system in parallel. This multi-lab validation demonstrated that the approach is robust, transferable, and suitable for a wide range of experimental questions.
While the method still has limitations – such working best at high cell density and the use of complex serum components – it nonetheless represents a major step towards more accessible stem cell research. By eliminating animal feeder cells, the system lowers technical and financial barriers and enables large-scale studies of early human development. “We’ve established a scalable, efficient, and developmentally faithful culture system”, Martin concludes. “Replacing serum with fully defined synthetic components will be the next critical step, as serum is a complex and variable mixture, and understanding its key components – such as relative importance of specific collagens and other critical serum components – will allow the development of standardized, animal-free culture systems for human stem cell research.
DOI: 10.1038/s44318-026-00714-2