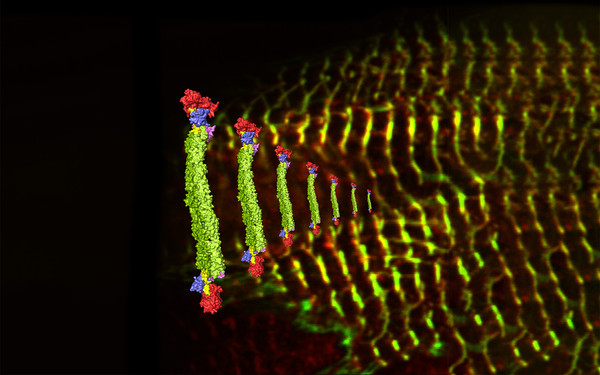
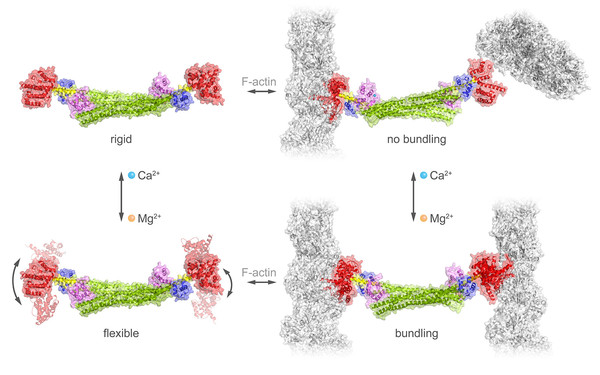
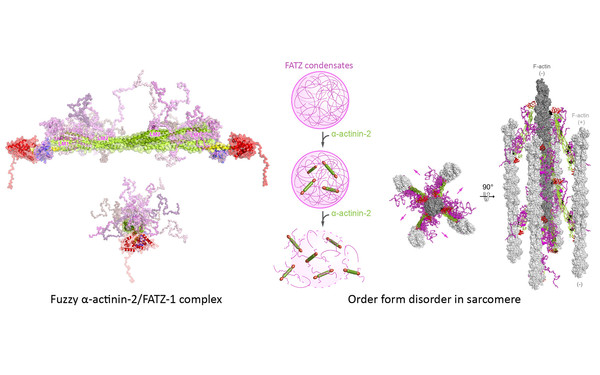
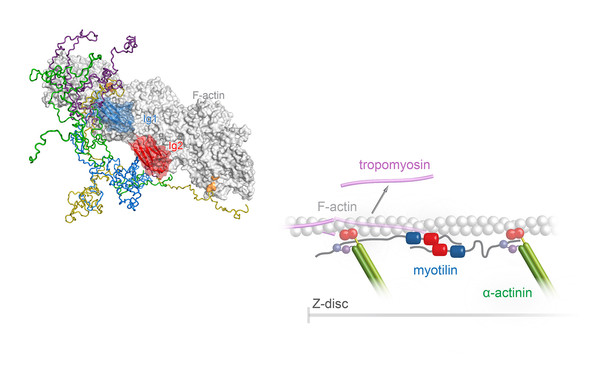
Member of Doctoral Program “Liquid-liquid Phase Separation in Biology – Ellipse”
Since 10/2021: Liquid-liquid Phase Separation in Biology – Ellipse, funded by the Austrian Science Fund FWF.
SLO FWF project I-5173-B (jointly with Brigita Lenarčič, University of Ljubljana)
Since 07/2021: Molecular Mechanisms of Calcium-Regulated alpha-Actinin, funded by the Austrian Science Fund FWF and Slovene Research Agency.
Member of Austrian-Slovak Interreg Project
(jointly with Eva Kutejova, Slovak Academy of Sciences)
Building, learning and research capacities in the structural and functional analysis of biomolecules for the needs of biomedicine and biotechnology. Total grant Volume: 1.166.798,81 €
05/19 - 04/22
http://www.imb.savba.sk/strubiomol/

Research Platform Comammox (jointly with Holger Daims, Michael Wagner, Kristina Djinovic-Carugo, and Andreas Richter, University of Vienna)
Since 6/2018: Exploring comammox: from molecules to ecology and applications, funded by University of Vienna.
http://www.microbial-ecology.net/news/new-comammox-research-platform-at-the-university-of-vienna
Vienna Science and Technology Fund (WWTF), Chemical Biology project LS17-008, (jointly with Christian Becker, Anne Conibear, Robert Konrat)
03/2018 – 08/2021: Structure ZOOM
Wellcome Trust Collaborative Award
2016-2020: An integrated approach to the muscle Z-disk: from atomic structure to human disease (with M. Gautel, P. Elliott, H. Watkins, S. Raunser and K Gehmlich)
Doctoral Program "Integrative Structural Biology"
2016-2019: The Group Djinovic participates in the special Doctoral Program "Integrative Structural Biology" reviewed and funded by the Austrian Science Fund FWF. Kristina Djinovic-Carugo is Vice Speaker of the program.
Doctoral Program "Signaling Mechanisms in Cellular Homeostasis"
The Group Djinović is an associated member of the special Doctoral Program "Signaling Mechanisms in Cellular Homeostasis" reviewed and funded by the Austrian Research Fund FWF.
Doctoral Program "Structure and Interaction of Biological Macromolecules"
2009-2013: Doctoral Program "Structure and Interaction of Biological Macromolecules" reviewed and funded by the Austrian Science Fund FWF.
COSS - Center for Optimized Structural Studies
2014-2016: Laura-Bassi Center of Expertise "COSS - Center for Optimized Structural Studies" funded by the Austrian Research Promotion Agency FFG (Co-ordinator)
2010-2013: Laura-Bassi Center of Expertise "COSS - Center for Optimized Structural Studies" funded by the Austrian Research Promotion Agency FFG (Co-ordinator)
Bioinformatics Integration Network III
2009-2013: Research Network funded by the GEN-AU Genome Research Program of the Federal Ministry of Science and Research
DFG/FWF Research Unit
2014-2017: "Structure and regulation of the Myofibrillar Z-disc Interactome" funded by the German Research Foundation and the Austrian Science Fund
2010-2014: Research Unit 'Alpha Actinin, Filamin C and their Complexes with Binding Partners' funded by the German Research Foundation and the Austrian Science Fund
FWF Stand Alone Projects
2014-2017: MORN “Structure and Function of the Trypanosoma brucei bilobe”
2010-2013: MORN repeat proteins and the Trypanosoma brucei bilobe (with G. Warren)
2010-2013: Structural Aspects of RAF-1:Rok-alpha Interaction (with M. Baccarini)