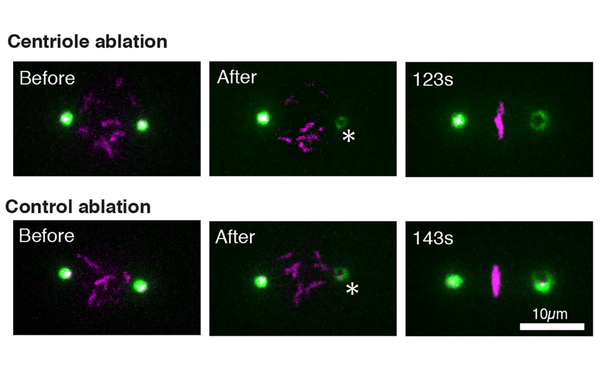
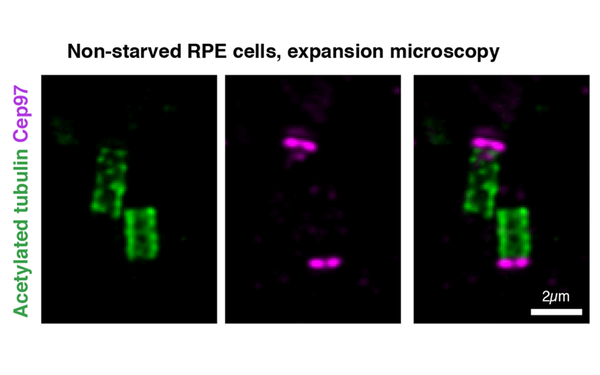
Centriole function in ciliogenesis
The other main function of centrioles, conserved in all species that possess them, is as basal bodies templating the formation of cilia. Ciliogenesis involves the docking of basal bodies to the plasma membrane and assembly of an elaborate gating structure, known as the transition zone, immediately distal to the basal body. Finally, the microtubule-based axoneme that serves the backbone of the cilium forms by direct extension of centriolar microtubules. Unusually, in C. elegans ciliated neurons, the basal body does not persist at the ciliary base, but degenerates during neuronal differentiation. Using a combination of light microscopy and electron tomography we showed that centrioles degenerate remarkably early in ciliogenesis, prior to expansion of the ciliary transition zone and axoneme elongation (Serwas et al., J Cell Biol 2017). Unlike in the case of centrosome assembly, centrioles are therefore required only for cilium initiation, but not directly involved in later steps of maturation and maintenance.