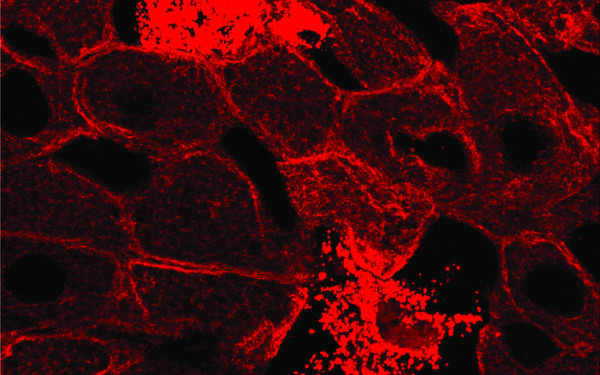
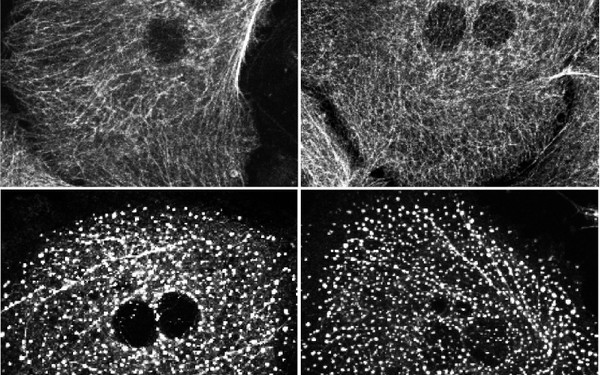
The Question
A major role of the keratin intermediate filaments in epithelia is to protect cells from mechanical and non-mechanical stresses. There is increasing evidence for the involvement of keratin-associated proteins with the modulation of these functions. One of these proteins is epiplakin, a member of the plakin protein family. We showed that in several liver stress models epiplakin-deficient mice suffered from aggravated liver injury, accompanied by increased formation of hepatocellular keratin granules. These data indicate that during certain cellular stress conditions, epiplakin aids in organizing the reassembly of new keratin filaments. How is epiplakin performing these tasks? By understanding the interactions of epiplakin with keratins in more detail, we expect to increase our knowledge about the mechanisms responsible for the formation of keratin filaments and their breakdown and to reveal potential correlations of epiplakin-mutations and pathologies of epithelial tissues and organs.