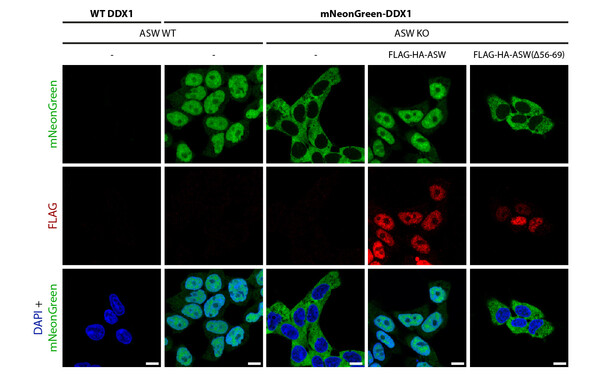
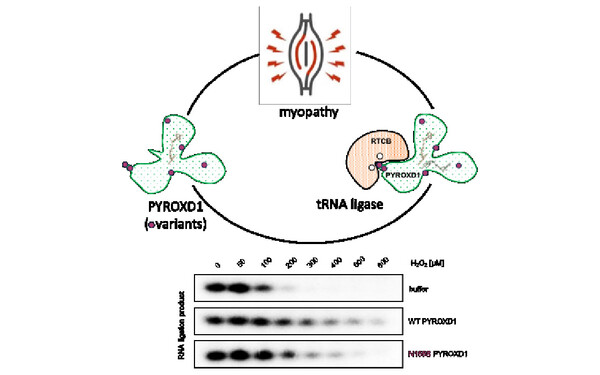
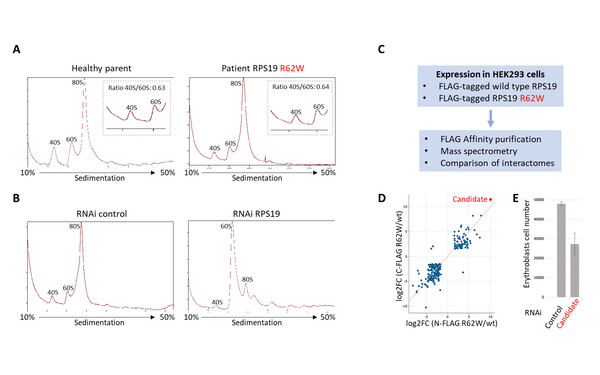
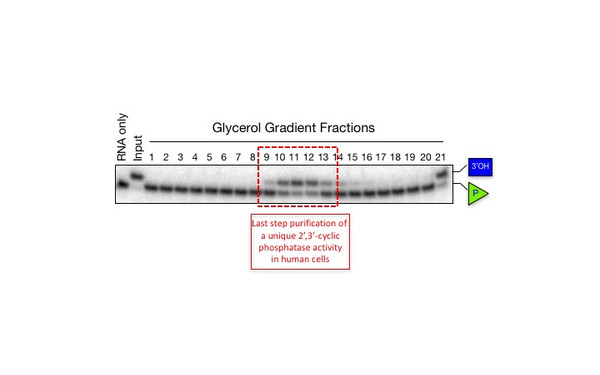
Javier Martinez
Javier Martinez obtained his PhD in Argentina, characterizing “cruzipain”, a major cysteine-proteinase in Trypanosoma cruzi. As a Post-Doc, he turned into RNA biology to identify the poly(A) ribonuclease PARN and the RNA-induced silencing complex – RISC – in human cells. As a Junior Group Leader at IMBA, Javier and his team discovered and characterized the RNA kinase CLP1 and the tRNA ligase complex, and these findings redirected his laboratory towards non-canonical RNA splicing, an essential pathway for the maturation of transfer RNAs. Javier is a Group Leader at the Max Perutz Labs and Professor of the Medical University of Vienna. Long time ago (!) – Javier used to be youth champion of table tennis in Argentina. He is also a football (River Plate, Barcelona and Manchester City) and a Formula 1 fan, but group meetings do not deal with these topics…